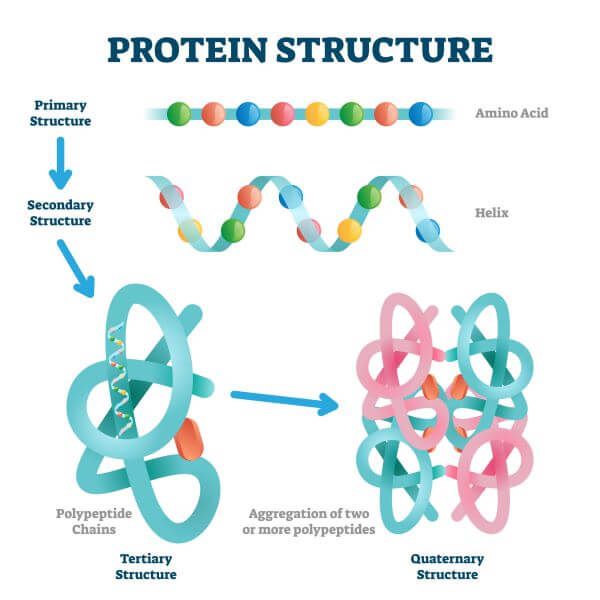
Peptide bonds are formed by a biochemical reaction that extracts a water molecule as it joins the amino group of one amino acid to the carboxyl group of a neighboring amino acid. Within a protein, multiple amino acids are linked together by peptide bonds, thereby forming a long chain. Secondary structure refers to highly regular local structures within a polypeptide (e.g., a helix) and either within or between polypeptides ( b-pleated sheets ).The building blocks of proteins are amino acids, which are small organic molecules that consist of an alpha (central) carbon atom linked to an amino group, a carboxyl group, a hydrogen atom, and a variable component called a side chain (see below). See 1972 Nobel Prize in Chemistry for more. Stein earned their share of the prize for relating the structure of the active site of the enzyme to its catalytic function). Christian Anfinsen won a half-share of the 1972 Nobel Prize in Chemistry for demonstrating that this was the case for the ribonuclease enzyme (Stanford Moore and William H. The higher order structures that account for the functional motifs and domains of a mature protein derive from its primary structure. In this way, primary structure reflects the genetic underpinnings of polypeptide and protein function. The unique physical and chemical properties of polypeptides and proteins are determined by their unique combination of amino acid side chains and their interactions within and between polypeptides. The different physical and chemical properties of amino acids themselves result from the side chains on their a-carbons. This is the same Sanger who first sequenced proteins, and yes…, he won a second Nobel Prize for the DNA sequencing work in 1980! Now, instead of directly sequencing polypeptides, we can infer amino acid sequences from gene sequences isolated by cloning or revealed after complete genome sequencing projects. The method of DNA sequencing, colloquially referred to as the Sanger dideoxy method, quickly became widespread and was eventually automated, enabling rapid gene (and even whole genome) sequencing. After transcription of a gene), a ribosome translates the resulting mRNA into a polypeptide.įor some time now, the sequencing of DNA has replaced most direct protein sequencing. Primary structure is dictated directly by the gene encoding the protein. By convention, the display and counting of amino acids always starts at the amino-, or N-terminal end (the end with a free NH2-group). Sanger received a Nobel Prize in 1958 for this feat. Each hydrolysis leaves behind a polypeptide fragment shortened by one amino acid that can be identified. Briefly, the technique involves stepwise hydrolysis of polypeptide fragments, called an Edman Degradation. Determining Protein Primary Structure - Polypeptide Sequencingįrederick Sanger was the first to demonstrate a practical method for sequencing proteins when he reported the amino acid sequence of the two polypeptides of Bovine (cow) insulin. You could prove this to yourself by assembling a short polypeptide with a molecular modeling kit, the kind you might have used in a chemistry class! The C-N-C-N-…backbone is the underlying basis of higher orders, or levels of protein structure (see below).ġ32 Amino Acid Sequence & Protein Primary StructureĢ. 
Amino acid side chains (circled above) end up alternating on opposite sides of a C-N-C-N-… polypeptide backbone because of the covalent bond angles along the backbone. The result of translation in a cell is a polypeptide chain with a carboxyl end and an amino end. A partial polypeptide is illustrated below. Just to make life interesting, L amino acids are actually dextrorotary in a polarimeter, making them d-amino acids! While both enantiomers exist in cells, only dextrorotary d (i.e., L) amino acids (along with glycine) are used by cells to build polypeptides and proteins. Recall also that only the lower case d or l defines the optical properties of isomers. Recall that chiral carbons allow for mirror image D and L or d and l optical isomers. 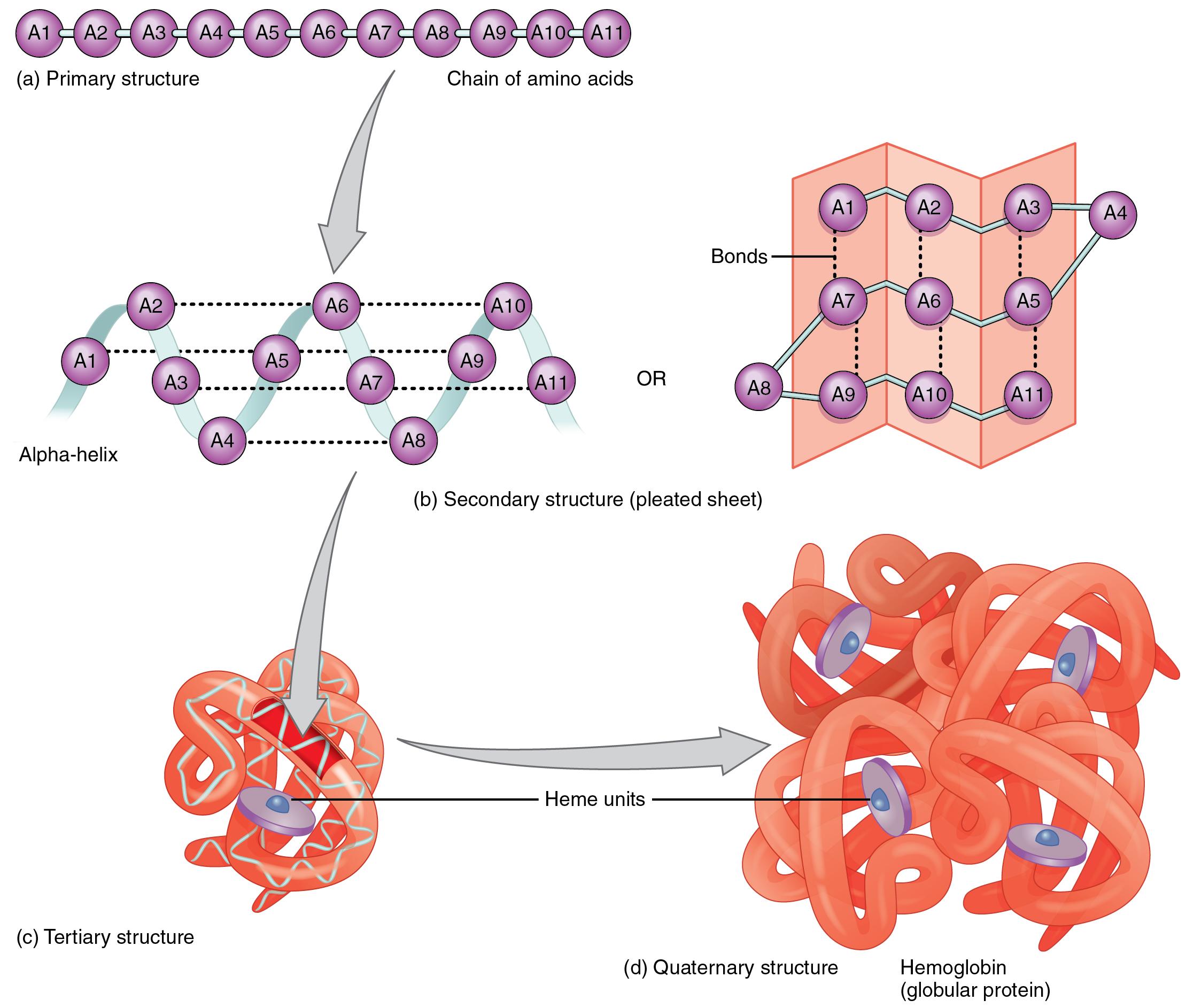
The 20 amino acids found in proteins are shown below.Įxcept for glycine, the a-carbon in the 19 other amino acids is bound to four different groups, making them chiral or optically active. They break and rearrange between the carboxyl and amino groups of amino acids during linkage formation. The linkages involve multiple covalent bonds. Peptide linkages between amino acids in polypeptides form in condensation reactions in cells during protein synthesis (i.e., translation). Cells use only 20 amino acids to make polypeptides and proteins, although they do use a few additional amino acids for other purposes. The primary structure of a protein refers to the amino acid sequence of its polypeptide chain(s). L amino acids and the C-N-C-N-… polypeptide backbone
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
